:strip_exif()/reboot/media/d17365d8-bdb1-11e8-beb0-fa163e14ea56/d1926104-bdb1-11e8-984a-fa163e14ea56/0-0-connaissances-sur-les-lampes-flash-au-xenon-knowledge-xenon-flashlamp.jpg)
Xenon Flashlamp for UV disinfection
Flashlamps are gas discharge devices filled with xenon gas and designe to produce pulsed radiation (from UV to IR). Flashlamp offers high efficiency and long lifetime (considering the lamp parameters). Flashlamp could be found in many sizes and shapes. The typical range of the plasma size is 20 to 250 mm but it could be higher with a competitive electronic !
Xenon gas
Introduction
Xenon was discovered by Ramsay and Travers in 1898. The name comes from the greek « xenos, which means « stranger ». Interested by the air composition, they used air liquid to make experiments. Once the air is transformed in liquid, it warms up. First, the nitrogen gas boils, then argon and finally oxygen.
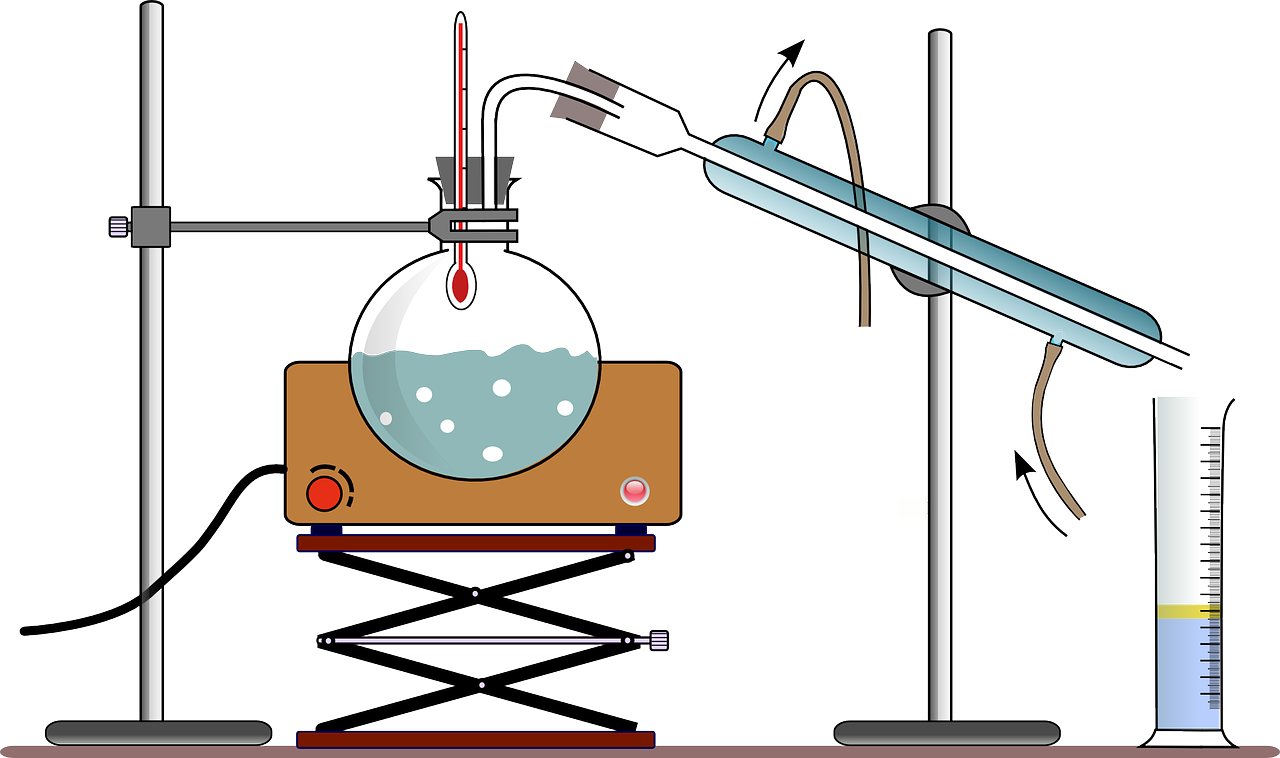
These three gases (nitrogen, argon and oxygen) form 99,95% of air.
But what about the 0,05% ?
Inside this 0.05 %, we find noble gases in very low concentration. One of the last gases to be isolated was Xenon. Noble gases remain in the air because they do not combine with other materials to form compounds (actually, this is what the researchers believed at this time).
Xenon gas is colorless, odorless, harmless, nonflammable, non toxic and inert chemically (or almost inert). Almost because an english chemist found way to make a xenon compound (XePtF6). To better understand the research, the best way is to look at this video : https://www.youtube.com/watch?v=Ejoct_6pQ74. In this video, an experiment is also realised to better understand the heaviness of the xenon noble gas.
Xenon in flashlamp
When a charge of electricity is passed through the gas, it emits a flash of blue light. Xenon is used when a very bright light is needed, close to the sun light. Xenon pulse uses the full spectrum to kill germs from UV (180 nm) to IR (1100 nm).
If the gas inside the lamp contains contamination, it could be a factor to explain a early end of life for the lamp.
Where does contamination come from ?
- Gas contamination could be the result of outgassing of electrodes (cathodes particularly). A correct fabrication of all the elements of the lamps is very important. If a manufacturing defect occurs, before filling gas, a contamination is possible. Nevertheless, in general, outgassing does not supply enough contamination to reduce the lifetime.
- Gas contamination could occur during the filling gas process and a small amount of air penetrates inside the lamp.
- Last possibility, gas contamination could come from the fused silica envelop. Due to the high energy, fused silica (SiO2) dissociates in Si (metal) and O2 (gas). The Oxygen gas contaminant inhibits rapidly triggering of the lamp. The lamp has then difficulty to start.
The step of filling gas must be definitely well performed to optimize the xenon lamp life time.
Envelop
The envelop is commonly synthetic fused silica. Synthetic fused silica (SiO2) has very low impurities, therefore, the transparency is better into the ultraviolet.

Natural fused silica is bannished for the disinfection application because of the impurities inside the material. Impurities inside the envelop could lead to solarization and a purplish discoloration occurs. Solarization is due to the formation of colored center in the presence of ion impurities. The consequence of solarization is the absorption of light and consequently, an increase in temperature of the silica material. And the lifetime of the xenon lamp may be reduced.
To conclude, if the application needs high level of UV light (as disinfection), synthetic fused silica will be requested.
Electrodes
Electrodes for xenon lamps must hold at extrem temperature, at high peak current (over 1000 A), over a long operating lifetime (over 12 millions of flashes). Generally, electrodes are made with tungsten or alloys of tungsten.
Electrode desintegration (particularly cathode) occurs due to pulsed discharges. Traces of metal are sputtered from the tip of the electrode to the fused quartz envelop. This phenomenon occurs very close to the electrodes. To reduce the sputtering, the best way is to discuss with the manufacturer about the operating conditions. Therefore, the designer will be able to adapt the shape of the electrode.
Moreover, xenon gas in contrast to other noble gas, reduce the sputtering rate because it provides a good transfer of heat and reduce the impact of the metallic evaporation. The sputtering rate could be also reduce by playing on the gas pressure. And finally, the sputtering rate is also dependent on purity of electrode material.
Conclusion
To get a correct functioning of the lamp, we should not skimp on the resources : Gas pressure, purity of the materials (electrodes, fused silica of the envelop), manufacturer with extensive experience, purity of the gas, shape of the electrode… Everything counts !
:strip_exif()/reboot/media/d17365d8-bdb1-11e8-beb0-fa163e14ea56/6bad1184-2d4c-11ef-80e8-0242ac120012/1-1-logo-sterixene-pulsed-light-et-uv-led-decontamination-par-lumiere-pulsee-desinfection-sterilisation.png)
:strip_exif()/reboot/media/d17365d8-bdb1-11e8-beb0-fa163e14ea56/6bad1184-2d4c-11ef-80e8-0242ac120012/1-1-logo-sterixene-pulsed-light-et-uv-led-decontamination-par-lumiere-pulsee-desinfection-sterilisation.png)
:recolor():strip_exif()/reboot/media/d17365d8-bdb1-11e8-beb0-fa163e14ea56/ebf8470e-180d-11ea-b407-0242ac130003/0-0-microbiological-tests.jpg)
:recolor():strip_exif()/reboot/media/d17365d8-bdb1-11e8-beb0-fa163e14ea56/7c41f174-4f46-11ea-817b-0242ac130005/0-0-interieur-machine-3.jpg)
:recolor():strip_exif()/reboot/media/d17365d8-bdb1-11e8-beb0-fa163e14ea56/77736ea4-180d-11ea-aae6-0242ac130003/0-0-biological-indicators.jpg)